


Dr. Ambika Ashraf
Director | Professor
Dr. Hussein Abdul-Latif
Professor
Dr. Joycelyn Atchison
Professor
Dr. Nicole Barnes
Assistant Professor
Dr. Giovanna Beauchamp
Assistant Professor
Dr. Christy Foster
Assistant Professor
Dr. Pallavi Iyer
Associate Professor
Dr. Kenneth McCormick
Professor
Dr. Gail Mick
Professor
Dr. Jessica Schmitt
Assistant Professor
Dr. Mary Lauren Scott
Associate Professor
Dr. Michael Stalvey
Associate Professor
Research from the UAB Division of Pediatric Endocrinology & Diabetes spans from the bench to the bedside. Through their research, division faculty and fellows strive to make an impact on the lives of patients. The division works in close collaboration with members of the Comprehensive Diabetes Center, Nutrition Obesity Research Center (NORC), Gregory Fleming James Cystic Fibrosis Research Center, and the UAB Center for Clinical and Translational Science.
The division’s research includes cystic fibrosis (CF), type 1 diabetes, type 2 diabetes, lipid disorders, lipoprotein metabolism, congenital hypothyroidism, thyroid nodules, thyroid and parathyroid disorders, polycystic ovary syndrome, non-alcoholic fatty liver disease, dietary intervention and epigenetics.
The research of Michael Stalvey, M.D., has parlayed from the translational models of cystic fibrosis (CF) to now include large-scale clinical studies. His sentinel paper describing improved growth in CF children treated with CF transmembrane conductance regulator (CFTR) correction has ignited the interest into the idea of an intrinsic defect in growth.
Dr. Stalvey is the national co-PI for the PROMISE Endocrine Sub-Study: “A Prospective Study to Evaluate Biological and Clinical Effects of Significantly Corrected CFTR Function (PROMISE).” This prospective, multi-center, observational study is designed to measure the clinical effectiveness of triple combination modulator therapy in individuals with cystic fibrosis (with one or more copies of the F508del mutation), assess salutary effects across a number of CF disease manifestations and collect specimens for future research. The major focus of the endocrine sub-study is to evaluate glucose metabolism, growth, bone metabolism and body composition in CF in response to the triple combination modulator therapy.
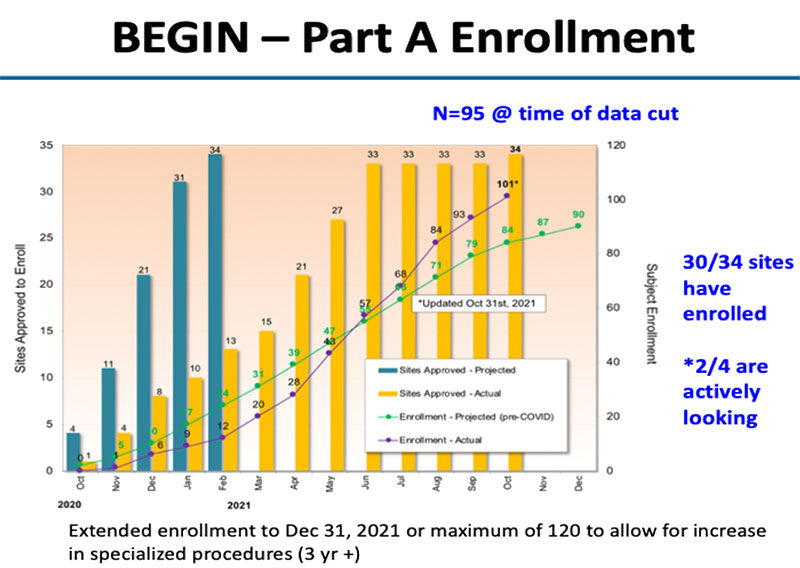
Dr. Stalvey recently launched a new study in infants and children with CF with support from the Cystic Fibrosis Foundation. He is co-principal investigator of “A Prospective Study to Evaluate Biological and Clinical Effects of Significantly Corrected CFTR Function in Infants and Young Children (BEGIN)” along with Bonnie Ramsey, M.D., Lucas Hoffman, M.D., Ph.D., and Sonya Heltshe, Ph.D. from the University of Washington and Seattle Children’s Hospital. This $11 million multi-center trial, including 30+ CF centers across the US, will coordinate the study of children with CF for the next seven years, setting the framework for research priorities for years to come. Dr. Stalvey’s team of investigators will collect natural history data in these very young CF children (less than six years), encompassing multiple facets of disease that are unique to children with CF at this age. The second part of the study will follow these young children as they go onto the triple combination modulator therapy and follow the implications on the comorbidities of early CF. The BEGIN study will set the stage for the next frontier—correcting CFTR function in infants and young children, hopefully delaying the onset or even preventing the most feared complications of CF.
The pathogenesis of type 1 diabetes mellitus (T1DM) involves autoimmune destruction of pancreatic beta cells leading to total insulin dependence. Gamma aminobutyric acid (GABA) is a common bioactive compound that has been shown to both prevent and reverse diabetes in animal models. Kenneth McCormick, M.D., led the first human trial of GABA in children with new onset T1DM. This was a Phase I, investigator-initiated, double-blind, placebo-controlled trial of low-dose GABA that enrolled 97 children age 4–18 years (clinicalTrials.gov NCT02002130). GABA was remarkably well-tolerated and had an excellent safety profile. Glucagon was significantly inhibited by GABA, and there was no change in circulating c-peptide. Data analysis continues on the pediatric GABA (gamma aminobutyric acid) and GABA/GAD (glutamate decarboxylase) clinical trial. The combination of GABA/GAD decreased fasting and meal-stimulated glucagon, confirming a pancreatic alpha cell action with potential therapeutic applications. Immunologic results from the GABA/GAD trial are forthcoming.
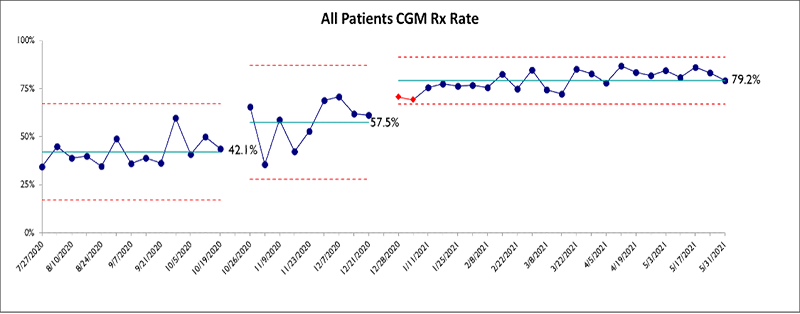
Ambika Ashraf, M.D., is collaborating with the Division of Pediatric Infectious Diseases (Claudette Poole, M.D., Swetha Pinninti, M.D., Suresh Boppana, M.D.) to determine the association of SARS-CoV-2 infection and comparative relative incidence of new-onset type 1 diabetes mellitus and type 2 diabetes mellitus (T2DM) in children in the state of Alabama. This study also aims to determine the association between virologic and antiviral antibody characteristics of SARS-CoV-2 infection in children with newly diagnosed T1DM and those presenting with diabetic ketoacidosis (DKA) and/or hyperglycemic hyperosmolar syndrome (HHS). Moreover, the study plans to determine the association between virologic and antiviral antibody characteristics of SARS-CoV-2 infection in children with newly diagnosed T2DM and those presenting with DKA and/or HHS.
Jessica Schmitt, M.D., and Dr. Ashraf are collaborating with Dr. Bisakha Sen at the UAB School of Public Health
to better understand the trajectory of incidence of pediatric patients with type 2 diabetes during the COVID-19 pandemic. They are reviewing the electronic health records and the Medicaid health claims data to quantify the rise in type 2 diabetes and delineate any ethnic or socio-economic disparities.
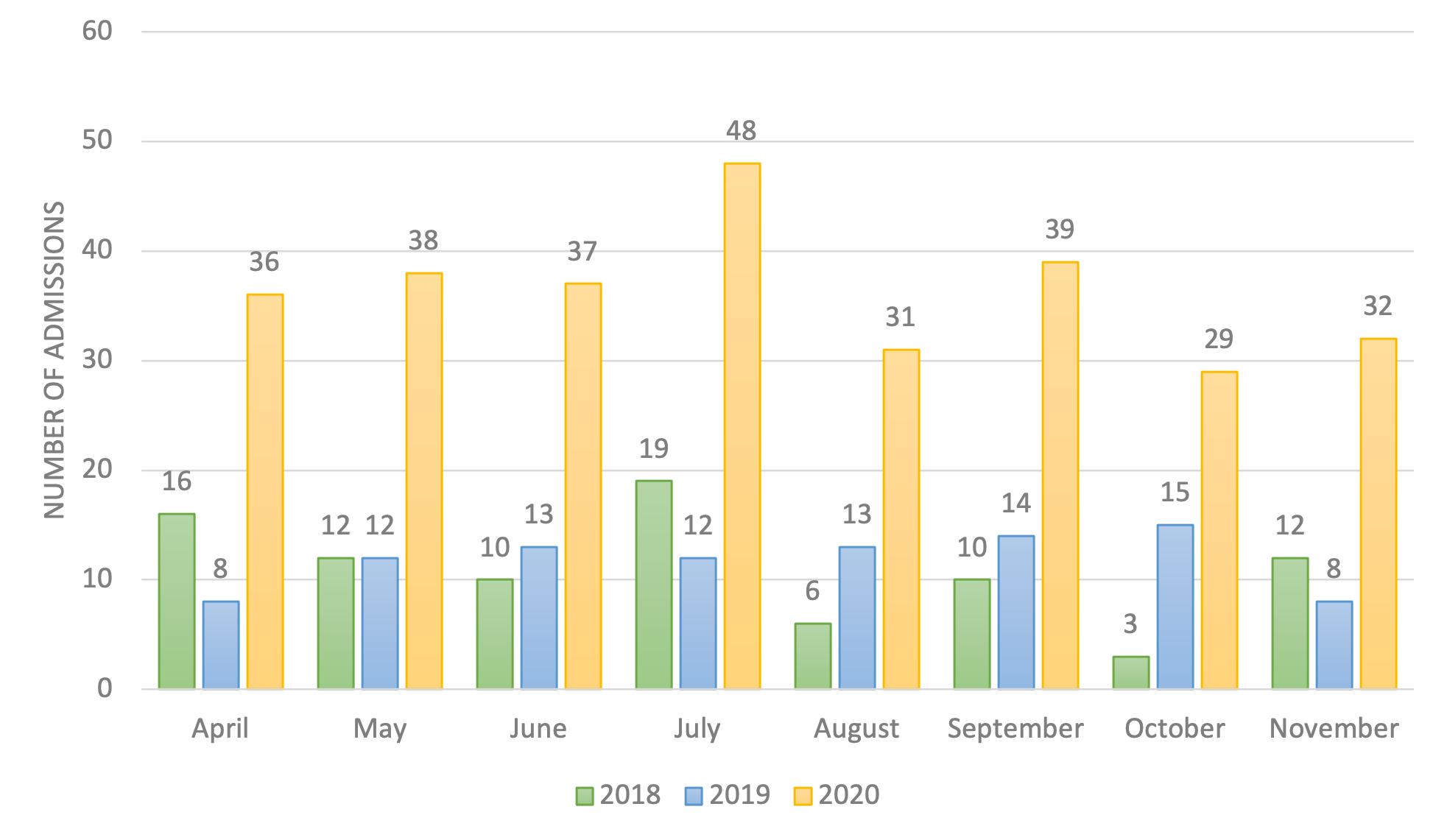
Dr. Ashraf, along with the researchers in the Division of Pediatric Infectious Diseases at UAB (Connie Trieu, Joshua Cooper, April Yarbrough, Swetha Pinninti, Suresh Boppana), evaluated the increase in the incidence of types 1 and 2 diabetes mellitus (T1DM, T2DM) between April–November 2020 at Children’s of Alabama. We observed a 16.3% increased rate of new onset T1DM and 205.3% increased rate of new onset insulin-dependent T2DM between April and November 2020 when compared to the same observational period in 2019. Among children with new-onset T1DM, 56.9% presented with DKA in 2019 and 47.1% in 2018 compared to 64.3% in 2020, which was higher than the national average. The study highlighted that the COVID-19 pandemic might have led to an increased rate of new onset T1DM, T2DM and DKA in children and adolescents compared to a similar period in the prior two years.
Figure 1. Hospitalized Children with New-Onset Type 1 Diabetes from April to November 2018–2020
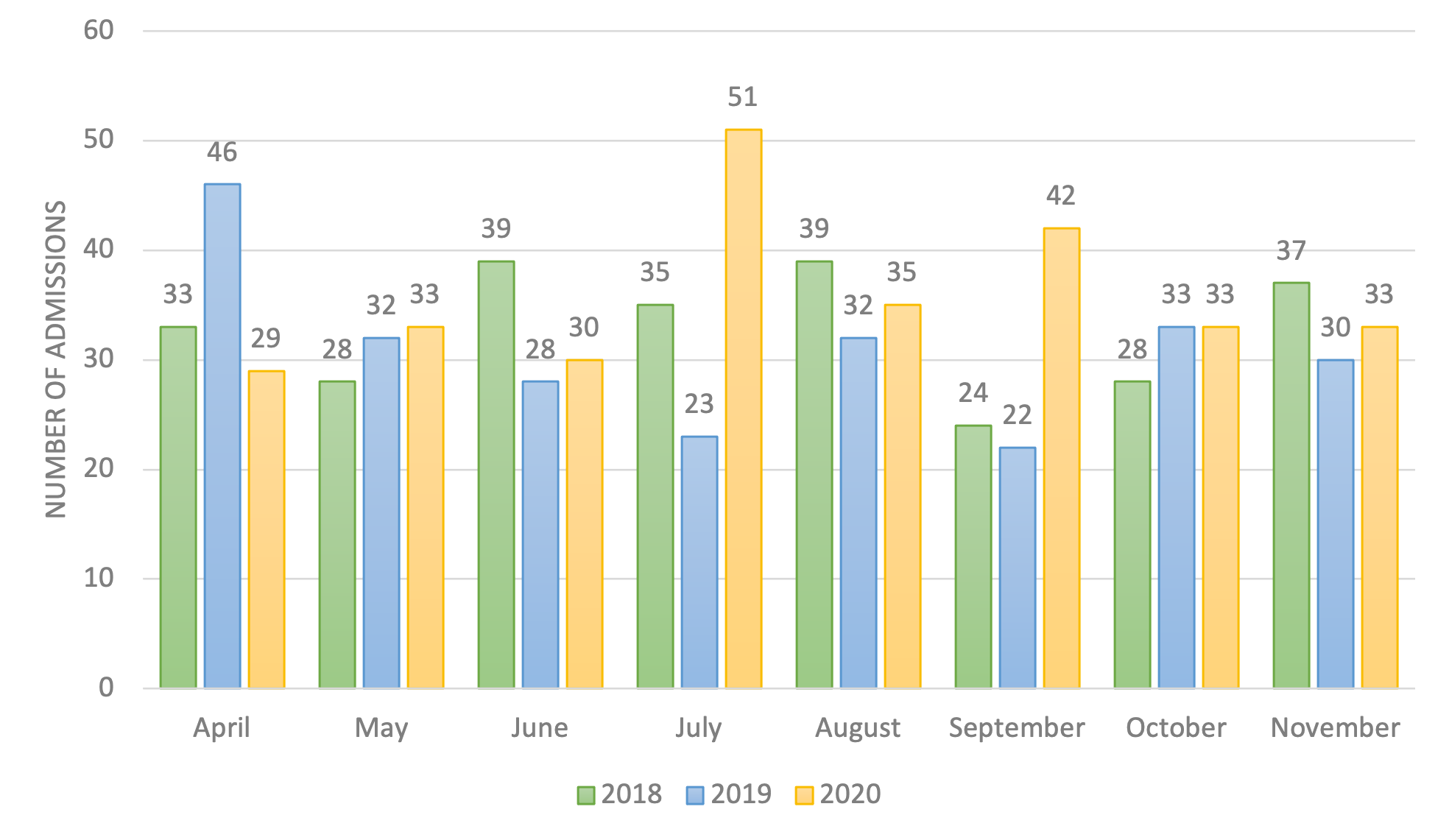
Figure 2. Hospitalized Children with New-Onset Type 2 Diabetes from April to November 2018–2020
Christy Foster, M.D., researches the understanding of the pathophysiology of polycystic ovary syndrome (PCOS), obesity and metabolic syndrome and how the associated comorbidities increase the metabolic risk of patients. Her current research involves evaluating DNA methylation (epigenetic) age. Relatively little is known about epigenetic age acceleration in adolescents with obesity, metabolic syndrome and type 2 diabetes, as this cohort continues to be an understudied population. The current study seeks for the first time to evaluate epigenetic age acceleration in adolescents with obesity and type 2 diabetes (T2DM). She hypothesizes that adolescents with obesity and T2DM will have an accelerated epigenetic age compared to their chronological age, and this acceleration will be higher than those with subjects without obesity and T2DM. She also hypothesizes systemic inflammation, early onset of menarche (before age 9), and poor glycemic control, commonly associated in adolescents with obesity and T2DM, will modify the epigenetic age acceleration age in these adolescents. This study will increase our understanding of epigenetic change in adolescents with obesity, thereby shedding light on the mechanisms of comorbidities that may occur with obesity later in life. Longer-term goals include conducting studies longitudinally and evaluating if interventions could alter the advanced epigenetic age. She hopes to build on the above research and study the use of pharmacotherapy in adolescents with obesity as well as its impact on the epigenetic age of adolescents.
Dr. Foster’s other clinical research area is in polycystic ovary syndrome, a condition of hyperandrogenism, dysfunctional ovulation and polycystic ovaries. To investigate potential genes, which could contribute to the development of PCOS, candidate gene studies were done. One candidate gene identified was DENND1A found on chromosome 2. Dr. Foster is working on investigating the amount of mRNA of DENND1A. V2 in the urine and serum is increased in adolescent females with PCOS. She is also actively involved in a multi-center collaboration in the creation of a database of adolescent females with PCOS. The database is titled the Presentation and Medical Course of PCOS in Adolescents Across the United States.
Ambika Ashraf, M.D., is investigating the role of gut microbiome in the pathogenesis of type 2 diabetes in children. The primary objective is to determine whether there are differences in gut microbiota in children with obesity, prediabetes and T2D contributing to the development of T2DM and response to treatment. The primary aim of this project is to quantify and evaluate the difference in the composition of gut microbiota between children with obesity, prediabetes and T2D. We will also evaluate temporal changes in the gut microbiome abundance and diversity. The study also intends to test whether baseline gut microbiota before treatment initiation for T2DM predicts successful response to treatment. Moreover, this project also compares the differences in lipoprotein profile and inflammatory marker concentrations before and after implementation of treatment. Medical students Charles Gagnon and Katherine Buchanan are actively involved in this project. Dr. Ashraf is collaborating with two renowned experts in the microbiome, Dr. Vivek Lal in the Division of Neonatology and Dr. Joseph Pierre at the University of Wisconsin.
Dr. Ashraf is the co-investigator along with Dr. Amy Goss from the UAB Nutrition Sciences Center, investigating the use of a carbohydrate-restricted diet as a treatment targeting normalization of hepatic lipid content in adolescents with non-alcoholic fatty liver disease. The study involves 12 weeks of controlled low-carbohydrate diet intervention study for the entire family, assessment of liver fat by MRI, metabolomics, euglycemic clamps, and then six months of “free living” dietary intervention.
Pallavi Iyer, M.D., is working to analyze genomic and RNA sequencing data from thyroid cancer surgical samples of children to find a molecular test that will help guide how best to care for children with thyroid nodules and thyroid cancer. Her research will detect gene alterations and fusion genes and correlate them with clinical findings. These novel markers could be used diagnostically for all children with suspicious thyroid nodules.
Hussein Abdul-Latif, M.D., is the site PI for multi-center research study using Carbitocin for children with Prader-Willi syndrome. The studies evaluate the safety and efficacy of the investigational drug Carbitocin as a potential novel treatment for Prader-Willi syndrome. Dr. Abdullatif is involved in an investigator initiative study assessing knowledge of families of children with type I on mini-dosing of Glucagon and level of interest in it in cases of hypoglycemia. Dr. Abdullatif also conducts studies on the use of burosumab in the treatment of nevus sebaceous associated hypophosphatemia. He is also conducting a study of perception of Meharry Medical students of their rotation at UAB and whether it affects their interest in applying for residency positions.
Alabama is one of 12 states that employs two newborn screens for the diagnosis of congenital hypothyroidism (CH) and congenital adrenal hyperplasia (CAH), with our state lab measuring both T4 and TSH on all CH screens. Continuing our commitment to early diagnosis and treatment of CH, Gail Mick, M.D., and Leslie Pitts CNRP are completing a three-year, follow-up analysis to determine whether lowering the statewide second screen TSH cutoff will detect previously undiagnosed infantile CH. An additional study in progress addresses the accuracy of neonatal thyroid ultrasounds in predicting later onset permanent CH. Dr. Mick is the site PI for an exciting Phase III, CAH trial examining the potential efficacy of a novel therapeutic agent that reduces excessive corticosteroid requirements by modulating corticotropin releasing factor.
Jessica Schmitt, M.D., collaborated with researchers from Children’s National Hospital, Loma Linda University Health, Children’s Hospital of Richmond, The University of Texas Health Science Center at Houston, and Cook Children’s Medical Center to evaluate the rate of central nervous system abnormalities found in children with isolated growth hormone deficiency. Dr. Schmitt, along with Falzur Rahman (UAB, Department of Biostatistics), was responsible for combined data analysis and interpretation. Results were published in the Journal of Pediatric Endocrine and Metabolism and support more judicious use of brain MRI in children with isolated growth hormone deficiency.
Am J Respir Crit Care Med. 2021 Nov 16. Clinical effectiveness of elexacaftor/tezacaftor/ivacaftor in people with cystic fibrosis. Nichols, D., Paynter, A., Heltshe, S., Donaldson, S., Frederick, C., Freedman, S., Gelfond, D., Hoffman, L., Kelly, A., Narkewicz, M., Pittman, J., Ratjen, F., Rosenfeld, M., Sagel, S., Schwarzenberg, S., Singh, P., Solomon, G., Stalvey, M., Clancy, J., Kirby, S., Van Dalfsen, J., Kloster, M., and Rowe, S.
J Clin Endocrinol Metab. 2021 Aug 7;dgab568. Case Studies in Pediatric Lipid Disorders and Their Management. Ambika P Ashraf, Bhuvana Sunil, Vaneeta Bamba, Emily Breidbart, Preneet Cheema Brar, Stephanie Chung, Anshu Gupta, Aditi Khokhar, Seema Kumar, Marissa Lightbourne, Manmohan K Kamboj, Ryan S Miller, Nivedita Patni, Vandana Raman, Amy S Shah, Don P Wilson, Brenda Kohn.
J Clin Transl Endocrinol. 2021:100271; SARS-CoV-2 infection in hospitalized children with type 1 and type 2 diabetes. Connie Trieu,Bhuvana Sunil; Ambika P. Ashraf; Joshua Cooper; April Yarbrough; Swetha Pinninti; Suresh Boppana.
J Cyst Fibros. 2021 Feb 19;S1569-1993(21)00038-2. PROMISE: Working with the CF community to understand emerging clinical and research needs for those treated with highly effective CFTR modulator therapy. Dave P Nichols, Scott H Donaldson, Carla A Frederick, Steven D Freedman, Daniel Gelfond, Lucas R Hoffman, Andrea Kelly, Michael R Narkewicz, Jessica E Pittman, Felix Ratjen, Scott D Sagel, Margaret Rosenfeld, Sarah Jane Schwarzenberg, Pradeep K Singh, George M Solomon, Michael S Stalvey, Shannon Kirby, Jill M VanDalfsen, John P Clancy, Steven M Rowe.
Ther Adv Endocrinol Metab. 2021 Nov 24;12:20420188211058323. Novel therapeutic targets and agents for pediatric dyslipidemia. Bhuvana Sunil, Christy Foster, Don P Wilson, Ambika P Ashraf.
Hussein Abdul-Latif, M.D.
Ambika Ashraf, M.D.
Pallavi Iyer, M.D.
Christy Foster, M.D.
Gail Mick, M.D.
Jessica Schmitt, M.D.
Mary Lauren Scott, M.D.
Michael Stalvey, M.D.

Nicole Barnes, M.D.
Assistant Professor
The UAB Pediatric Endocrinology Fellowship Program consists of a three-year comprehensive experience that incorporates clinical care, research activities and evidence-based learning.
Our ACGME-accredited program accepts a total of six fellows. Our curriculum is a balance of inpatient and outpatient clinic rotations, with ample time devoted to developing research and/or career interest. Clinical management of outpatient and inpatient endocrine disorders is the emphasis of the first year of training. The second and third years are devoted to pursuing research opportunities in an area of basic or clinical research under the supervision of a faculty preceptor. Fellows are encouraged to participate in other institutional clinical experiences here at UAB, including genetics, reproductive endocrinology and adult endocrinology. Research experiences are available through our division or vast resources here at UAB. These research opportunities may include basic science, clinical or transitional research, as well as projects in medical education.
In addition to diabetes and general endocrine clinics, we participate in multi-disciplinary clinics, such as Turner Syndrome clinic, Metabolic Bone Disorders clinic, Gender dysphoria, Obesity clinic and CFRD clinics. Other subspecialty clinics include Newborn Screen clinic, High-risk Diabetes clinic, Type 2 Diabetes clinic, PCOS, Metabolic Bone Disorders clinic, Thyroid Nodule clinic, and Lipid clinics. We offer monthly educational activities that include journal clubs, board prep didactics and questions, “Lunch and Learn” with faculty and quarterly career prep sessions. Fellows are encouraged to participate in other Institutional clinical experiences at UAB, including exposure to clinical genetics, reproductive endocrinology, adult endocrinology and endocrine surgery. Research experiences are available through our division or vast resources at UAB. These research opportunities may include basic science, clinical or transitional research, as well as projects in medical education or quality improvement. Our institution participates in industry studies, the Pediatric Diabetes Consortium and T1D Exchange. We provide funding support for those interested in pursuing a master’s degree during fellowship training, as well as funding for subspecialized certificates in the areas of obesity and lipids, among others.
While UAB Pediatric Endocrinology has experienced significant growth, we pride ourselves on still offering our fellows a “small program” feel with their training. It is, in our opinion, the best mix of large clinical volume and specialization without losing our personal touch and connection with the fellows. We consider ourselves a family and are committed to nurturing our trainees to be the best pediatric endocrinologists they can be.
2021–2022 Fellows
Whitney Smith, M.D., Ph.D.
Second-Year Fellow
Residency: University of Mississippi
Margaret Marks, M.D.
Third-Year Fellow
Residency: University of Alabama at Birmingham
Leen Matalka, M.D.
Third-Year Fellow
Residency: University of Alabama at Birmingham
Program Directors
Mary Lauren Scott, M.D.
Program Director
Giovanna Beauchamp, M.D.
Associate Program Director
Program Coordinator
Toni Davison