The UAB Department of Pediatrics established the Pediatric Research Office (PRO) in 2015 to renew its commitment to the generation of new knowledge in the diagnosis, treatment and sequelae of pediatric diseases. The PRO seeks to “lower the energy of activation” in the design, conduct and analysis of research conducted within the Department of Pediatrics. The success of the PRO is reflected in the departmental accomplishments outlined in Dr. Cohen’s introduction to this year’s Academic Annual Report. In FY 2021, the Department of Pediatrics had funding from the National Institutes of Health (NIH) totaling over $21.5 million and total research funding of $33.5 million, reflecting a 21% increase in NIH funding and 17% increase in overall funding compared with last year. NIH-funded investigators have increased from 21 to 27 this year, and 24 of these investigators have used PRO services to advance their research.
The PRO is led by David W. Kimberlin, M.D., Vice-Chair for Clinical and Translational Research in the Department of Pediatrics. PRO personnel and associated partners provide assistance with the following core components of clinical and translational research:
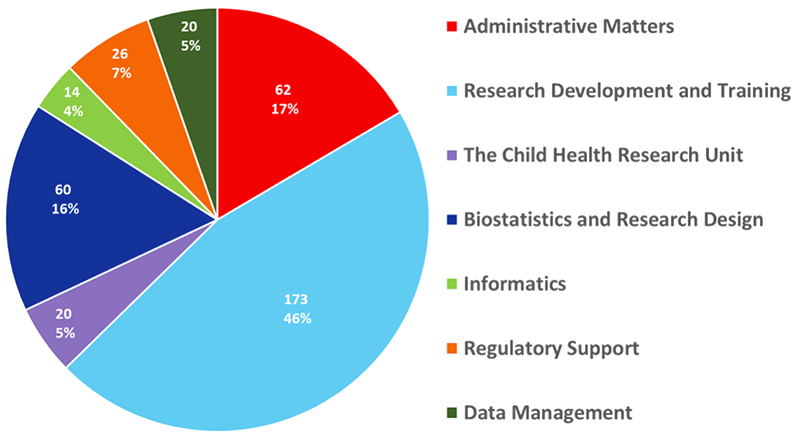
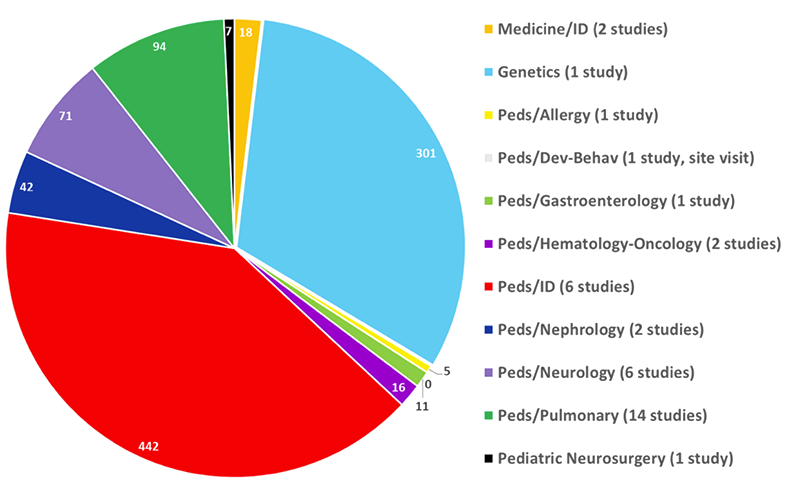
From October 2020 through September PRO PROJECTS BY TYPE OF USER, 10/20–9/21 (N=375) 2021, the PRO assisted investigators and their study teams with 361 projects that included 375 services (projects can have more than one service if, for example, one project requested help with both biostatistics and informatics). Help was provided to 181 unique users (e.g., faculty fellows, residents, etc., as shown in the chart). Services were used by 90% of the Department of Pediatrics’ divisions, as well as by those doing pediatric research within 13 other departments, including: Anesthesiology, Child and Adolescent Psychiatry, Dermatology, Epidemiology, Genetics, Medicine, Microbiology, Neurobiology, Neurology, Neurosurgery, Obstetrics and Gynecology, Psychology and Surgery.
As noted above, the PRO manages the primary Child Health Research Unit (CHRU), which is a partnership between the UAB Department of Pediatrics, Children’s of Alabama and the UAB Center for Clinical and Translational Science (CCTS). It provides outpatient space for pediatric research to reduce barriers to the conduct of scientifically rigorous clinical and translational research. The primary CHRU opened in 2017 in a renovated, 2,547-square-foot facility on the third floor of Dearth Tower. This space includes a reception/registration area, a triage room, six well-equipped exam rooms, two conference rooms, workspace with monitors and locked storage, a lab with centrifuge and freezer for short-term storage, and an equipment storage room. There is also a CHRU Ancillary Unit on the seventh floor, which is used primarily for pulmonary research. This Unit has three exam rooms, a conference room, work areas for study teams, ECG and pulmonary equipment, refrigerated centrifuges, specimen storage, sterile hoods, a 40x-100x microscope, a hemocytometer and an autoclave.
CHRU use was reduced in early 2021 due to the COVID-19 pandemic. However, two COVID vaccine trials (sponsored by Pfizer and Moderna) resulted in an overall increase in visits of 106% (462 visits in FY20 to 950 visits in FY21). From 10/1/20 to 9/30/21, the main CHRU and the Satellite Unit combined were used by 23 unique investigators for 37 studies.